Please click on the link below to download the Other R&D Pipeline | Black Morel Mycoprotein Project PDF.
Please click on the link below to download the Immunomodulatory Effects of Fungal β-glucans PDF.
Please click on the link below to download the β-glucans Metabolic and Immunomodulatory Properties and Potential for Clinical Application PDF.
Please click on the link below to download The Effects of C. lacerata on Insulin Resistance in Type 2 Diabetes Patients PDF.
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
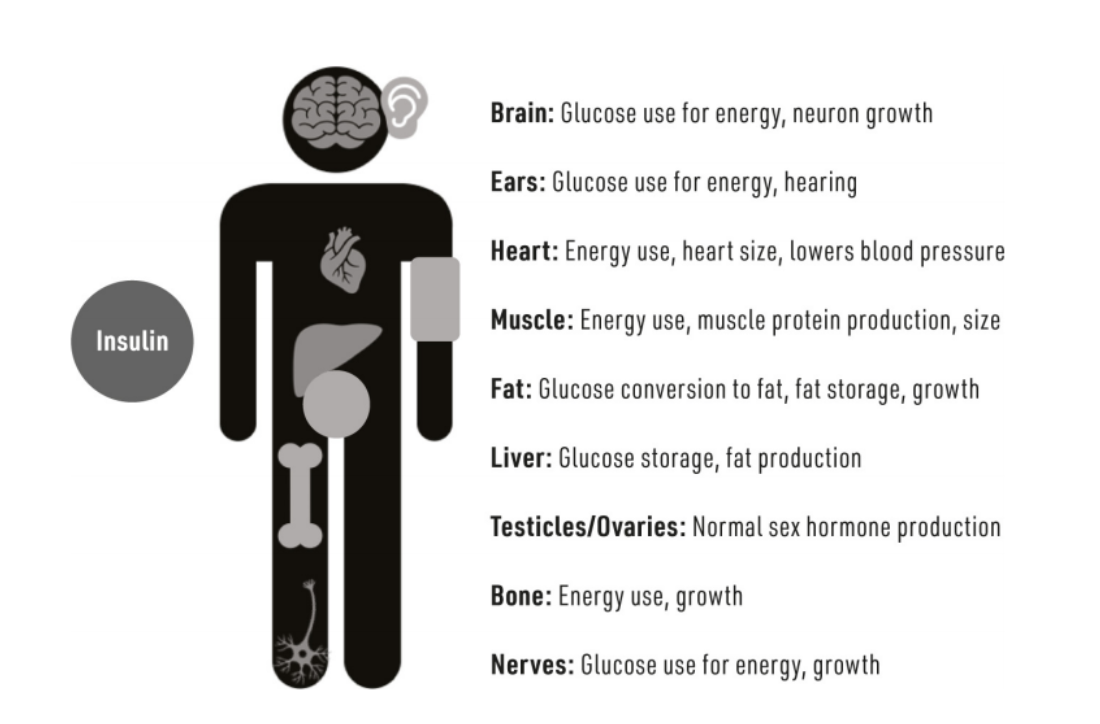
Insulin, Insulin is a hormone produced by the beta cells of the pancreas. insulin influences every cell in every tissue of the body. Insulin resistance is a root cause of numerous chronic diseases, including cardiovascular disease, cancer, cerebrovascular disease, and diabetes.
It acts as a "glucose transporter," crucial for moving glucose from the blood into cells to generate energy. All carbohydrates break down into glucose in the blood; insulin facilitates glucose uptake by cells.
Insulin serves as a significantly better predictor of type 2 diabetes than glucose, potentially identifying at-risk individuals up to 20 years before the onset of symptoms.
Reference:APA 7th Edition:
Groopman, G. M. (2007). Why we get sick: The hidden epidemic of treatment-resistant disease and how it’s transformed modern medicine. Houghton Mifflin Harcourt.
Beta Cell Dysfunction and Insulin Resistance
Please click on the link below to download the full version of the paper
Click to download
Diabetes Discovery Platform, South African Medical Research Council, Cape Town, South Africa
Edited by: Soroku Yagihashi, Hirosaki University Graduate School of Medicine, Japan
Reviewed by: Hiroki Mizukami, Hirosaki University Graduate School of Medicine, Japan; Naoto Kubota, University of Tokyo, Japan
Correspondence: Marlon E. Cerf, Diabetes Discovery Platform, South African Medical Research Council, PO Box 19070, Tygerberg, Cape Town 7505, South Africa. e-mail: marlon.cerf@mrc.ac.za
The study on beta cell dysfunction and insulin resistance provides an in-depth analysis of their respective roles in the pathogenesis of type 2 diabetes mellitus (T2DM). The findings emphasize that while both conditions contribute to hyperglycemia, beta cell dysfunction serves as a primary determinant of disease progression.
1. Beta Cell Dysfunction
2. Insulin Resistance
3. The Interplay Between Beta Cell Dysfunction and Insulin Resistance
Clinical Implications
o GLP-1 receptor agonists and DPP-4 inhibitors enhance beta cell insulin secretion and survival.
o SGLT2 inhibitors reduce glucotoxicity by promoting urinary glucose excretion, indirectly preserving beta cell function.
o Anti-inflammatory strategies may mitigate cytokine-induced beta cell apoptosis.
Conclusion
This study highlights beta cell dysfunction as the primary driver of type 2 diabetes pathogenesis, with insulin resistance acting as an exacerbating factor. Understanding their intricate interplay provides a foundation for targeted therapeutic approaches aimed at delaying or preventing the onset of diabetes.
Therapeutic Potential and Mechanisms of Action of Ceriporia lacerata
Introduction
The era of cell nutrition intervention for the prevention and treatment of chronic diseases has arrived. We are a biotechnology company registered in Singapore. After 20 years of research by our scientific team, we have discovered and proven the health benefits of the Ceriporia lacerata strain series for human health. Building upon the initial discovery, we developed the second-generation strain Ceriporia lacerata K1 and the third-generation strain Irpex Lacteus f.k. We have already conducted animal experiments on toxicity and safety tests using Ceriporia lacerata K1 fermented raw material EFILA, achieving breakthrough progress and positive market feedback. In summary, based on the research conducted by scientists from various fields, we have summarized and published the following research findings.
Ceriporia lacerata is a type of white-rot fungus that has gained considerable attention for its potential therapeutic applications in various biological systems, particularly in metabolic and immune health. Recent research has highlighted its beneficial effects on diabetes, insulin resistance, immune function, and cognitive function. This review synthesizes findings from multiple studies to present an overview of the biological effects and mechanisms underlying the therapeutic potential of C. lacerata.
1. Metabolic and Hypoglycemic Effects
The early studies on Ceriporia lacerata focused on its effects on glucose metabolism and insulin sensitivity. The submerged culture of C. lacerata mycelium has demonstrated hypoglycemic effects, as evidenced by Shin et al. (2015), who reported the C. lacerata mycelium's potential to lower blood glucose levels, which is crucial for the management of type 2 diabetes. This finding was further corroborated by Kim et al. (2023), who conducted a clinical trial investigating the anti-diabetic efficacy of the submerged culture medium of C. lacerata. They observed significant improvements in glycemic control in diabetic patients, suggesting its potential as a complementary treatment for diabetes.
Moreover, several studies have focused on the insulin signaling pathways. In the 2016 study by Shin et al., C. lacerata mycelium was shown to influence the insulin signaling pathway in 3T3-L1 cells, a model for adipocyte differentiation and insulin sensitivity. This effect on insulin signaling is significant for addressing insulin resistance, a hallmark of type 2 diabetes. Additionally, C. lacerata increased GLUT4 protein expression in db/db mice (a model for obesity and type 2 diabetes), which is crucial for glucose uptake into cells and further suggests that the fungus enhances insulin sensitivity.
A key mechanism identified in these studies is the modulation of PI3K/PKB (also known as Akt) signaling. The study by Kim et al. (2013) showed that the crude extract of C. lacerata has a protective effect on dexamethasone-induced cytotoxicity in INS-1 cells, likely through the activation of PI3K/PKB activity. This pathway is known to promote cell survival and metabolic processes, making it a critical target in the prevention and treatment of metabolic diseases like diabetes.
2. Immune-Enhancing Effects
Apart from its metabolic benefits, C. lacerata has been shown to have immune-enhancing properties. In a study by Hwang et al. (2022), the submerged culture of C. lacerata was found to improve immune function in cyclophosphamide-induced immunosuppressed mice. The underlying mechanisms appear to involve the activation of macrophages, key cells in the immune response. This suggests that C. lacerata may hold promise in modulating immune function, which could have therapeutic implications for conditions like autoimmune diseases or immune suppression caused by chemotherapy.
3. Cognitive and Neuroprotective Effects
Recent studies have expanded the therapeutic potential of C. lacerata beyond metabolic and immune functions, suggesting its neuroprotective and cognitive-enhancing properties. Lee et al. (2021) demonstrated that dietary supplementation with C. lacerata improved learning and memory in a scopolamine-induced amnesia mouse model, which is widely used to study cognitive impairment. This indicates that C. lacerata could have potential as a natural treatment for conditions such as Alzheimer's disease or other forms of dementia.
4. Conclusion and Future Directions
In conclusion, Ceriporia lacerata shows significant promise as a natural therapeutic agent for a range of conditions, including diabetes, insulin resistance, immune deficiencies, and cognitive impairments. The effects of C. lacerata appear to be primarily mediated through its influence on insulin signaling, glucose transport, immune modulation, and possibly through direct neuroprotective mechanisms.
Despite the encouraging preclinical and clinical findings, further research is required to fully elucidate the active compounds responsible for these effects and to confirm their efficacy in larger, more diverse human populations. Additionally, the long-term safety and potential side effects of C. lacerata supplementation must be thoroughly investigated before it can be recommended as a mainstream treatment.
In the future, C. lacerata could be integrated into functional foods or dietary supplements aimed at improving metabolic health, boosting immune function, or protecting cognitive function. Moreover, studies that explore its interaction with conventional drugs used for diabetes or immune disorders could lead to novel combined therapeutic strategies.
Thus, Ceriporia lacerata represents a fascinating area of research with promising therapeutic potential, particularly in the fields of metabolic and immune health, as well as cognitive enhancement.
Ceriporia lacerata 的治疗潜力与作用机制
引言
细胞营养干预在慢性疾病的预防与治疗领域的时代已经到来。我们是一家注册于新加坡的生物技术公司,在经过科学团队20年的研究后,我们发现并证实了Ceriporia lacerata菌株系列对人体健康的益处。基于最初的发现,我们开发了第二代菌株Ceriporia lacerata K1和第三代菌株Irpex Lacteus f.k.。我们已经使用Ceriporia lacerata K1发酵原料EFILA进行动物毒性与安全性测试,并取得了突破性进展和积极的市场反馈。总而言之,基于来自多个领域科学家的研究,我们总结并发布了以下研究成果。
Ceriporia lacerata 是一种白腐菌,因其在多种生物系统中的潜在治疗应用,特别是在代谢和免疫健康方面,受到广泛关注。近期的研究突出了它对糖尿病、胰岛素抵抗、免疫功能和认知功能的有益作用。本综述汇总了多项研究的结果,展示了Ceriporia lacerata的生物学效应和其治疗潜力背后的机制。
1. 代谢与降糖作用
最早的研究集中在Ceriporia lacerata对葡萄糖代谢和胰岛素敏感性的影响。Ceriporia lacerata的沉淀培养菌丝展示了降糖作用,Shin等人(2015)报告称,Ceriporia lacerata菌丝有可能降低血糖水平,这对2型糖尿病的管理至关重要。Kim等人(2023)进一步通过临床试验研究了Ceriporia lacerata沉淀培养基对糖尿病的抗糖尿病疗效,结果显示糖尿病患者的血糖控制有显著改善,表明其作为糖尿病辅助治疗的潜力。
此外,多个研究集中在胰岛素信号传导途径上。Shin等人(2016)的研究显示,Ceriporia lacerata菌丝可影响3T3-L1细胞的胰岛素信号通路,该细胞模型用于研究脂肪细胞分化与胰岛素敏感性。这种对胰岛素信号的影响对解决2型糖尿病的胰岛素抵抗问题具有重要意义。此外,Ceriporia lacerata还在db/db小鼠(肥胖和2型糖尿病模型)中增加了GLUT4蛋白的表达,GLUT4对葡萄糖进入细胞至关重要,进一步表明该真菌有助于提高胰岛素敏感性。
这些研究揭示的关键机制之一是PI3K/PKB(也称为Akt)信号通路的调节。Kim等人(2013)的研究显示,Ceriporia lacerata的粗提取物对地塞米松诱导的INS-1细胞毒性具有保护作用,这可能通过激活PI3K/PKB活动来实现。该通路已知可以促进细胞存活和代谢过程,是预防和治疗如糖尿病等代谢性疾病的关键靶点。
2. 免疫增强作用
除了代谢益处外,Ceriporia lacerata还表现出了免疫增强作用。Hwang等人(2022)的研究发现,Ceriporia lacerata的沉淀培养物可以改善环磷酰胺诱导的免疫抑制小鼠的免疫功能。其作用机制似乎涉及巨噬细胞的激活,巨噬细胞是免疫反应中的关键细胞。这表明,Ceriporia lacerata在调节免疫功能方面可能具有潜力,对于如自身免疫性疾病或化疗引起的免疫抑制等情况具有治疗前景。
3. 认知与神经保护作用
近期研究将Ceriporia lacerata的治疗潜力扩展到代谢和免疫功能之外,揭示了它的神经保护和认知增强特性。Lee等人(2021)的研究表明,食用Ceriporia lacerata能够改善由东莨菪碱诱导的健忘小鼠模型中的学习和记忆,这一模型广泛用于研究认知障碍。这表明Ceriporia lacerata可能成为治疗阿尔茨海默病或其他形式痴呆症的天然疗法。
4. 结论与未来方向
总之,Ceriporia lacerata作为一种天然治疗剂,在糖尿病、胰岛素抵抗、免疫缺陷和认知障碍等多种疾病中的潜力显著。Ceriporia lacerata的作用主要通过影响胰岛素信号、葡萄糖转运、免疫调节以及可能的直接神经保护机制来实现。
尽管初步的临床前和临床研究结果令人鼓舞,但仍需要进一步的研究来全面阐明这些效果背后的活性化合物,并确认它们在更大、更具多样性的人群中的疗效。此外,必须彻底研究Ceriporia lacerata补充剂的长期安全性及潜在副作用,才能在临床中广泛推荐使用。
未来,Ceriporia lacerata可作为功能性食品或膳食补充剂的一部分,旨在改善代谢健康、增强免疫功能或保护认知功能。此外,探索其与常规用于治疗糖尿病或免疫疾病的药物的相互作用可能会导致新型联合治疗策略的出现。
因此,Ceriporia lacerata代表了一个充满潜力的研究领域,特别是在代谢、免疫健康以及认知增强领域。
References:
lease click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download
Please click on the link below to download the PDF.
Click to download