Longrunn Medical Technologies was established in Singapore. Our team possesses over 20 years of research experience and is committed to developing natural, safe, and effective products for diabetic patients aimed at the prevention and treatment of diabetes and liver diseases. We have pioneered the development of several novel microbial strains utilizing our proprietary synthetic biotechnology, grounded in the principles of Cellular Nutrition. Our approach focuses on fungal fermentation as a strategic pathway for drug development. In 2025, EFILA’s core active ingredient and Blood Sugar Support formulation were officially recognized by the Singapore Health Sciences Authority (HSA), further strengthening our scientific and regulatory foundation.
At Longrunn Medical Technology, we believe that nature offers us an invaluable wealth of natural medicines.By integrating cutting-edge biotechnology with our deep respect for natural resources, we aim to deliver scientifically validated and genuinely effective health products, particularly for patients with diabetes.
Our mission is not merely to provide products, but to inspire hope and improve lives through the power of nature.


We are dedicated to becoming a global leader in the prevention and treatment of diabetes. By integrating innovative research, advanced biotechnology, and a deep understanding of natural medicinal resources, we aspire to provide cutting-edge solutions that empower individuals in their health journeys.
Our goal is to set new standards in diabetes management through evidence-based products and holistic approaches, ultimately improving the quality of life for millions around the world.
First Discovery of the Strain : This unique strain, discovered on Korean Pine Tree medicinal mushrooms, has demonstrated excellent efficacy in lowering blood sugar levels and addressing diabetes at its root cause—pancreas degeneration.
 2005
2005 Conducted 1st human clinical trial to evaluate the efficacy
 2008
2008 Obtained Food Safety Certification: Conducted GLP (toxicity test and efficacy test) and study commissioned by South Korea government
 2011
2011 Published four papers, focusing on national research projects designated by the Korean government.
 2013
2013 Completed a phase of human clinical trials, and the ingredient was approved by the KFDA as a health food for blood sugar regulation
 2019
2019 Certificate of Patent in South Korea
 2022
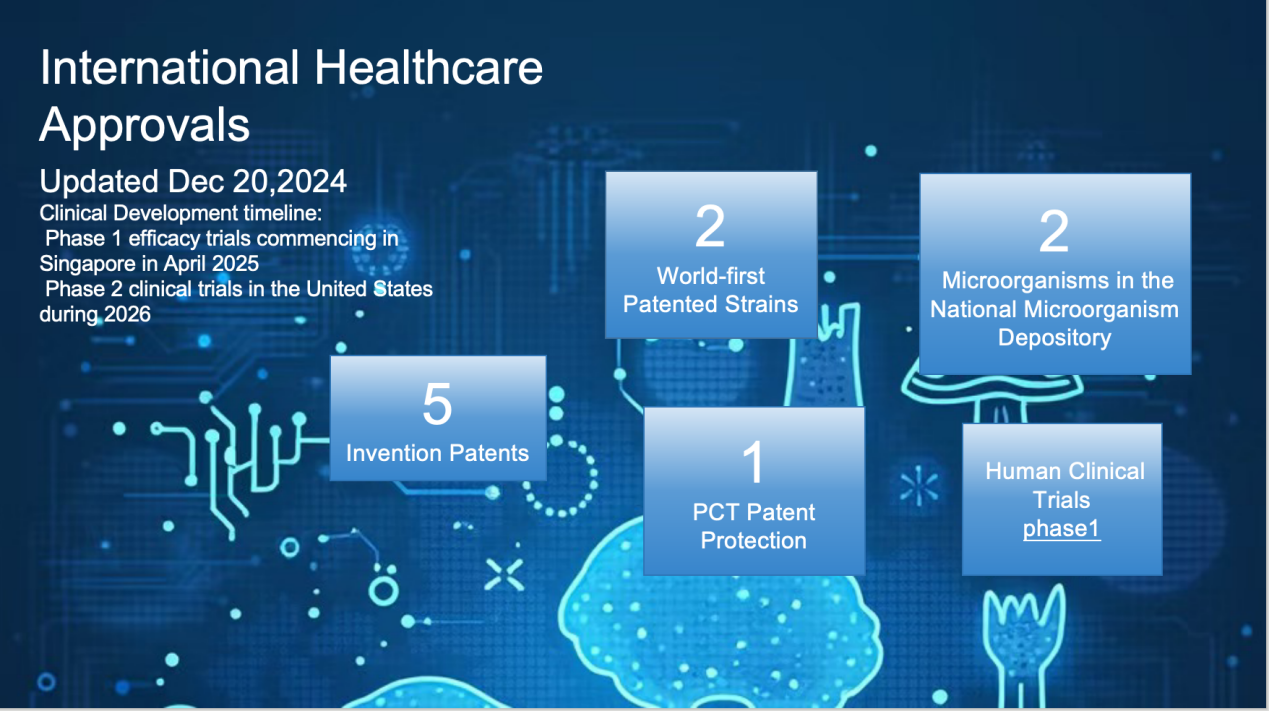
2022 Obtained five invention patents and PCT patent protection, as well as certification from the Singapore Health Sciences Authority (HSA).
 2024
2024