The Story of EFILA

EFILA (commonly referred to as EFILA Multi-Target Cellular Nutrition Intervention) is a cutting-edge health intervention solution that integrates synthetic biology and cellular medicine. It primarily targets chronic metabolic diseases, particularly Type 2 Diabetes Mellitus (T2DM) and Non-Alcoholic Fatty Liver Disease (NAFLD, now widely known as MASLD).
Its core concepts and technical features can be summarized in the following key dimensions:
Core Mechanisms: Multi-Target & Cellular Nutrition
Unlike traditional medicine, which often focuses on a single target (such as a single receptor or enzyme), EFILA emphasizes "multi-target synergy."
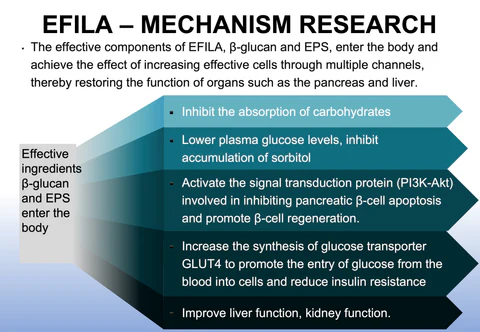
Cellular-Level Repair: Rather than relying solely on exogenous blood sugar reduction, it directly acts on damaged pancreatic \beta-cells and hepatic cells through highly bioavailable cellular nutrients.
Reversing Mitochondrial Dysfunction: It optimizes mitochondria—the cell's energy powerhouse—to reduce oxidative stress and chronic inflammation.
Improving Insulin Resistance: By repairing cell membrane receptors and intracellular signaling pathways, it restores the body's sensitivity to insulin.
Technical Foundation: Driven by Synthetic Biology
The underlying R&D of EFILA relies heavily on synthetic biology technologies:
High Purity & High Bioactivity: Utilizing engineered microorganisms or biocatalytic techniques, it bio-synthesizes specific nutritional components (such as specific small-molecule peptides, high-purity plant extract derivatives, or cytokine precursors) with maximum purity and bioactivity.
Overcoming Absorption Barriers: Through precise molecular structure design, it enhances the stability and intestinal absorption rate of these ingredients, ensuring targeted delivery to key organs (e.g., the liver and pancreas).
R&D Background & International Regulatory Approvals
The solution is backed by a strong international research background and cross-border regulatory compliance:
Joint R&D Background: Supported by core academic and technology transfer expertise from A*STAR (Agency for Science, Technology and Research), Singapore's premier research institution.
Dual-Country Regulatory Approval: It has secured regulatory approvals from health and food authorities in both Singapore and South Korea, granting it legal compliance for international commercialization and clinical nutritional intervention.
Commercial & Clinical Positioning
WHAT IS DIABETES?
Most patients are not aware that high blood sugar is just a symptom of diabetes and not the disease itself. Diabetes is a result of the body not producing enough insulin to metabolise the sugar we consume, or the body is resistant to insulin and thus the sugar is unable to be absorbed by the cells to be converted to energy.
Taking the commonly prescribed medicine only helps with improving the symptoms of diabetes, but not directed at the condition itself.
Ceriporia Lacerata was discovered in August 2005 by Korean scientist Dr. Kim Byoungcheon during his research on the functional substances contained in medicinal mushrooms. Dr Kim has a MBA from Kyungpook National University and a phD from Western California Medical University in the USA. He has also written multiple research paper after spending 20 years researching, studying and conducting clinical trials on this now patented ingredient. This unique strain, found on Korean Pine Tree medicinal Mushrooms, demonstrated excellent efficacy against lowering of blood sugar and targeting DIABETES from the root cause of pancreas degeneration. Other than the achievement of being awarded the Invention Patent for the manufacturing method of Ceriporia Lacerata culture broth extracts and pharmaceutical composition for prevention and treatment of Diabetic Disease and diabetic complications, he has also personally been given the prestigious Grand Award by both the World Inventor's Association and also in the World Invention Innovation Contest. He also won the gold medal in the International Art-Creativity & Engineering Exhibition as well as in the EURO INVENT 2015.

Using a patented fermentation technology, Dr Kim cultivated the patented micro-organism strain Irpex Lacteus FK extracted from medicinal mushrooms found on Korean Pine Trees. This strain was also isolated and deposited with the International Patent Micro-organism Depository Institution: the Microbial Bank of the National Agricultural Science Institute in Korea in 2021.

In early 2022, we obtained a patent for the Invention and Utilization of Metabolite EFILA derived from this strain for the prevention and treatment of various disease.
EFILA began its research and development in 2005, going through processes such as strain discovery, strain cultivation, fermentation product cultivation, product development and manufacturing, animal experiments, human trials, clinical trials, etc. This has formed the entire industry chain from strains to raw materials, and from raw materials to products.
Since the beginning of the research, there has been:
- 4 general toxicity tests
- 3 genetic toxicity tests
- 2 efficacy tests
- 3 safety pharmacology tests.
The roadmap for EFILA is to first enter the market as a medical-grade dietary supplement and then develop into auxiliary drugs for the prevention and treatment of diabetes and liver diseases.

Patient blood sugar levels after consuming
EFILA BLOOD SUGAR SUPPORT
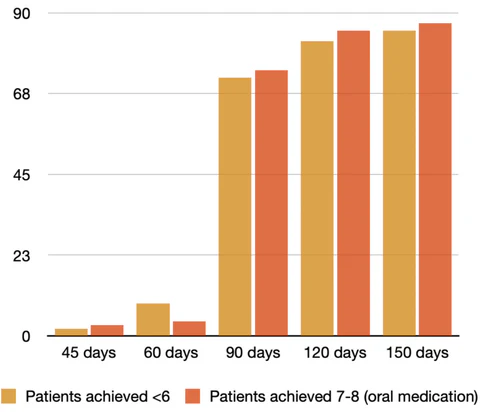
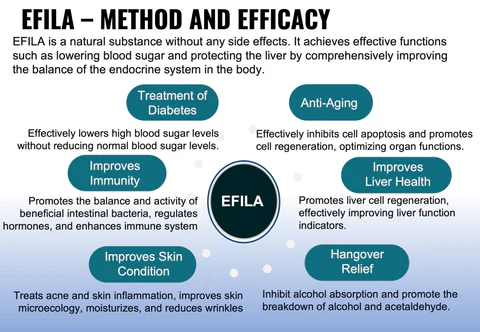
Through clinical studies, EFILA has been proven to have significant positive impacts on the prevention and treatment of diabetes and liver diseases. EFILA is mainly applied in the prevention and treatment of diabetes and liver diseases. Users have also reported positive effects on eyesight, skin, digestion, constipation and sleep. Efila can prevent aging in all aspects of the human body.
Longrunn Medical will continue to research and develop EFILA as a leader in cellular nutrition and create more beneficial supplements targeting at conditions associated to insulin resistance and liver deficiency.

Longrunn Medical x A*STAR: Pioneering Natural Solutions for Diabetes Care
Longrunn Medical Technologies is proud to announce a strategic collaboration with Singapore’s Agency for Science, Technology and Research (A*STAR) under the T-UP Programme.
Research Focus
This one-year project investigates the antidiabetic potential of Ceriporia lacerata K1 (CLK1), a rare white-rot fungus with unique bioactive compounds. The research will focus on:
-
Large-scale cultivation of CLK1 and extraction of active metabolites
-
Safety and efficacy validation through cytotoxicity and antidiabetic assays
-
Elucidating molecular mechanisms (e.g., PI3K/Akt, AMPK pathways)
-
Identifying and characterizing active antidiabetic compounds within CLK1, and advancing them into consumer-ready formats such as dietary supplements, functional foods, and complementary medicines
Strategic Significance
Through this partnership, Longrunn contributes expertise in nutraceutical innovation, while A*STAR brings world-class scientific research capabilities. Together, we aim to provide safer, natural, and effective solutions for diabetes management.
This collaboration not only addresses one of Singapore’s most pressing health challenges but also strengthens the nation’s position as a hub for translational health research and nutraceutical innovation.

EFILA ADVANCE BLOOD SUGAR SUPPORT — Officially Listed by HSA Singapore
- Approved Ingredient: Submerged Culture of Ceriporia lacerata mycelium
- Notified Product: EFILA ADVANCE BLOOD SUGAR SUPPORT
Key Milestones
• July 2025 — HSA (Ms. Kwok) confirmed approval of our new active ingredient request, officially recognizing “Submerged culture of Ceriporia lacerata mycelium” under the Voluntary Notification System (VNS).
Note: The culture medium alone requires a separate safety submission.
• September 2025 — HSA (Ms. Ria) completed document verification and confirmed that EFILA ADVANCE BLOOD SUGAR SUPPORT is included in the List of Notified Health Supplements and Traditional Medicines.
• VNS Number: VNS-2025-07-00175
Significance
• Official HSA recognition of Ceriporia lacerata as a functional, safe, and verified ingredient in Singapore.
• Validates EFILA’s scientific and regulatory strength in product formulation and compliance.
• Enhances EFILA’s brand credibility and supports future expansion across the Southeast Asian market.




